
Cian Hughes • Patient Advisory Board Member of Tinnitus Quest
Foreword
At Tinnitus Quest, one of our core principles is the belief that patients must be an integral part of driving the research agenda. No one understands the urgency of solving tinnitus better than those who live with it every day. For this reason, ensuring that patients have a meaningful voice at every step has become foundational to how we operate as an organization.
Our first major step towards finding a solution that could silence tinnitus came in 2025, with the opening of our inaugural grant application process in July. In line with our patient-driven philosophy, the Patient Advisory Board played an active role in reviewing proposals and contributing to decision-making, bringing lived experience and deep personal motivation into the evaluation process.
Our First Impression of the Research Proposals
By the time the application window closed in September, we had received 29 proposals from research groups around the world. These covered a wide range of approaches, including neuromodulation and brain stimulation, cochlear and auditory nerve-focused treatments, pharmaceuticals and other compounds, and cross-modal interventions targeting different neural circuits. Overall, there was a healthy mix of treatment modalities, many aligned with current directions in tinnitus research, and it was encouraging to see such global commitment to advancing the field.
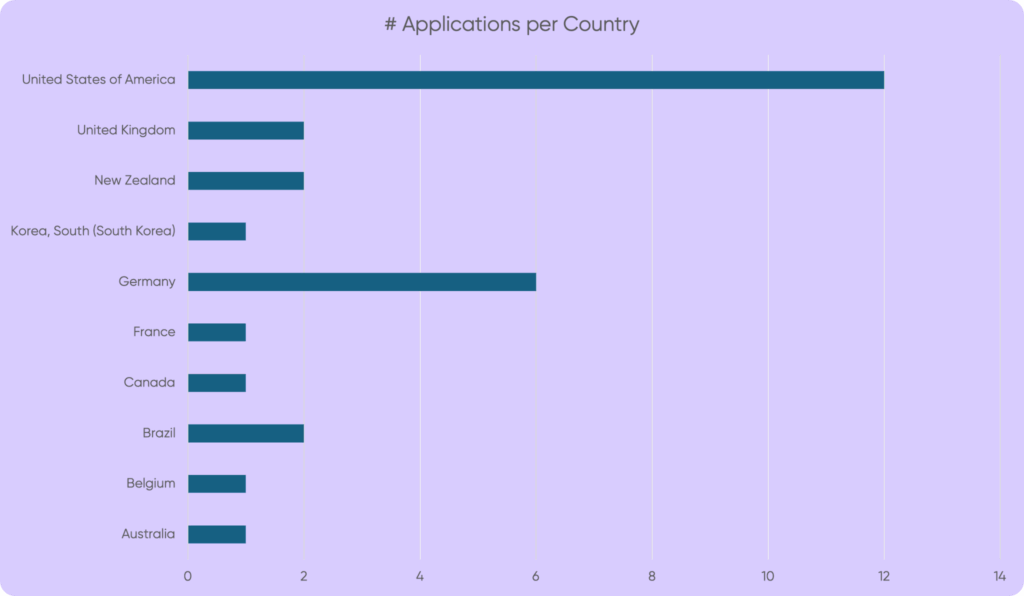
That said, I did feel some disappointment at the general lack of true innovation. Few proposals explored genuinely novel mechanisms or bold methodological departures, with many refining or modestly extending existing approaches rather than opening new conceptual directions. To me, this underscored the importance of initiatives like the Tinnitus Quest Hackathon held in October 2025, which created space for researchers to step outside traditional silos and begin exploring high-risk, high-reward ideas with transformative potential.
How We Assessed the Proposals
The Patient Advisory Board began its work with an independent review by each of the five members. We focused on research objectives, scientific rationale, outcome measures, and alignment with Tinnitus Quest’s mission. As patients, our central question throughout was simple but fundamental: Which of these projects truly has the potential to silence tinnitus? We also considered how quickly a treatment might reach patients, how accessible it could be, and whether there were risks of adverse effects. Each member then ranked the proposals according to their preference for advancement to the live pitch stage.
We subsequently came together for an extended Patient Advisory Board meeting to compare rankings, identify areas of agreement, and discuss differences. Over several hours, we worked systematically through every application, sharing what we saw as strengths and voicing concerns. Some proposals were quickly discounted by unanimous agreement, typically where previous research had shown no benefit or where the scientific rationale felt weak.
Encouragingly, our top choices were largely aligned across the board. While there were a few edge cases that required deeper discussion and advocacy, this debate was constructive and ultimately strengthened our confidence in the final outcome. By the end of the meeting, we had agreed on eight proposals, ranked in order, to recommend for the next stage. These spanned neuromodulation, sensory and deep brain stimulation using vibrotactile, electrical, and ultrasound approaches, as well as neurotrophins — reflecting a diverse but promising set of strategies.
In parallel, Tinnitus Quest’s Innovation Board conducted its own independent review and ranking of the proposals, submitting their assessments to the Executive Board, which made the final decision on whom to invite to live pitch meetings. Ultimately, six applicants were selected to advance. Their proposals included neuromodulation, non-invasive deep brain stimulation, trigeminal nerve stimulation, and biologic compounds aimed at repairing the auditory nerve.
The Patient Board was pleased to see that four of our eight recommendations made it into this final group. This alignment with leading tinnitus researchers on the Innovation Board was deeply validating and reinforced the credibility and impact of a patient-led review process.

Meeting the Applicants
The live pitch meetings took place over two weeks in early November and were attended by representatives from the Executive, Innovation, Scientific Advisory and Patient Advisory Boards. Each one-hour session began with a 15-minute “elevator pitch” presentation by the applicant, followed by 45 minutes of questions.
Core topics included the tinnitus mechanisms being targeted, expected effects on tinnitus loudness versus habituation, potential adverse events, and pathways to patient access. Additional questions sought to clarify feasibility and risk. After each applicant left the meeting, the Tinnitus Quest representatives held a brief internal debrief to share impressions.
From a patient perspective, these meetings were both exciting and hopeful. It was heartening to see serious research efforts underway around the world aimed at tackling tinnitus. At the same time, the live pitches sometimes changed my perception of proposals — occasionally for the better, but in other cases raising new doubts after hearing how researchers justified their rationale and addressed concerns.
Looking Forward…
Following the pitch meetings, further reflection and re-review led to additional questions for some applicants, which were resolved via email to ensure full clarity and transparency.
Ultimately, Tinnitus Quest awarded one grant, to the University of Oxford, for a study testing transcranial ultrasound stimulation of deep brain regions to reduce tinnitus loudness. From the outset, the Patient Advisory Board was impressed by the proposal’s novelty and scientific rigor. We recommended increasing the sample size and incorporating minimum masking level measurements to assess tinnitus loudness. Our suggestions were welcomed by both the Executive Board and the researchers themselves.
Looking ahead, this grant marks an important milestone and sets the stage for an exciting year. Throughout 2026, we will share regular updates on the progress and impact of the funded research – starting around the summer, when the Oxford project will kick off, after the researchers get approval from the university’s Ethics Committee (a hard requirement for any testing on humans).
As such, we will keep our community closely connected to the work they support. We also plan to launch our next grant cycle earlier in the year, aiming for April 2026, allowing us to build momentum, engage researchers sooner, and continue pushing towards meaningful breakthroughs in tinnitus research.













2 Comments
Thank you for sharing this, you guys are amazing. Would you be able to talk a little more about the application that tried to treat the auditory nerve with neutrophins?
Mine is driving me to lunacy.